
With that size, it takes over 18 million of these atoms, lined up side by side, to equal the width of your little finger (about 1 cm). Individual atoms are extremely small even the largest atom has an approximate diameter of only 5.4 × 10 −10 m. In Chapter 1 "Chemistry, Matter, and Measurement", we defined an atom as the smallest part of an element that maintains the identity of that element. He was a physicist from the country of Denmark. The modern atomic theory The fundamental concept that all elements are composed of atoms., proposed about 1803 by the English chemist John Dalton ( Figure 2.2 "John Dalton"), is a fundamental concept that states that all elements are composed of atoms. Why niels bohr is important Niels Bohr is important for his work in the areas of atomic structure and quantum theory. But how far can you take this exercise, at least in theory? Can you continue cutting the aluminum foil into halves forever, making smaller and smaller pieces? Or is there some limit, some absolute smallest piece of aluminum foil? (Thought experiments like this-and the conclusions based on them-were debated as far back as the fifth century BC.) All attention is focused on the explanation of concrete phenomena. Atomism - Modern Theory, Particles, Structure: With the development of a scientific atomic theory, the general philosophical problems gradually disappeared into the background. It should be obvious that the pieces are still aluminum foil they are just becoming smaller and smaller. To a certain extent modern atomic theory has bridged the gap between atomistic and holistic thought. Continue cutting, making smaller and smaller pieces of aluminum foil. Cut one of those smaller pieces in half again. Now you have two smaller pieces of aluminum foil. Explain how all matter is composed of atoms.answer the question why is the atomic theory important, which will help you get the most accurate answer. Through different experiments with gases Dalton expanded on this to theorise that atoms vary in size and mass and that compounds had to be made of whole number ratios of atoms. You are wondering about the question why is the atomic theory important but currently there is no answer, so let summarize and list the top articles with the question. This built on the work of Lavoisier and Dalton furthered this proposing that each chemical element is made of atoms of a unique type and they cannot be altered or destroyed but can be combined. He found that 100g of tin will combine with 13.5g or 27g of oxygen and that this could be represented by a 2:1 ratio, for every 2 atoms of oxygen there was one atom of tin. He looked at tin oxide and the combination of masses of oxygen with tin.  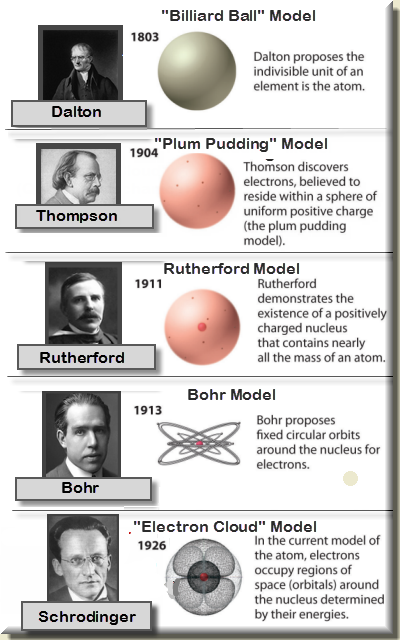
Dalton used the work of Lavoisier and Joseph Proust to examine the ratios of elements that combine to form compounds and look at their ratio of masses. The work of Lavoisier and the atomists was furthered in the 18th Century by the British scientist John Dalton. It also proved the earlier work of Robert Boyle who hypothesised in 1661 that elements cannot be broken down into simpler substances. This was a crucial breakthrough in the work of atomists in confirming what matter was made of as it was proved that atoms are not created or destroyed when a reaction happens. This led to the theory of the law of conservation of mass. They formulated the key concepts of the law of conservation of mass and the existence of atoms as the building blocks of all matter using their knowledge of chemical reactions.Ī later breakthrough in the discovery of the atomic model came through the work of French chemist Antoine Lavoisier who through a series of experiments found that the total mass of products and reactants in a chemical reactions is always the same. Home Science Physics Matter & Energy Development of atomic theory The concept of the atom that Western scientists accepted in broad outline from the 1600s until about 1900 originated with Greek philosophers in the 5th century bce. For example, if an atom has a Z of 6, it is carbon, while a Z of 92 corresponds to uranium. The modern Atomic Model was first developed by two key scientists Lavoisier and Dalton with the help of others. The single most important characteristic of an atom is its atomic number (usually denoted by the letter Z), which is defined as the number of units of positive charge (protons) in the nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
